Focal Adhesion Kinase (FAK) is an un-receptor-specific protein of 125 kDa Tyrosine Kinase, which acts as an Src substrate. Src is a crucial component for integrin signaling.
FAK is an essential component in cell spread cell differentiation, cell death, migration, and the acceleration of cell death and acceleration of the G1 transition from the G1 to S change in cell cycles. You can know more about Anti-FAK/PTK2 Antibody online.
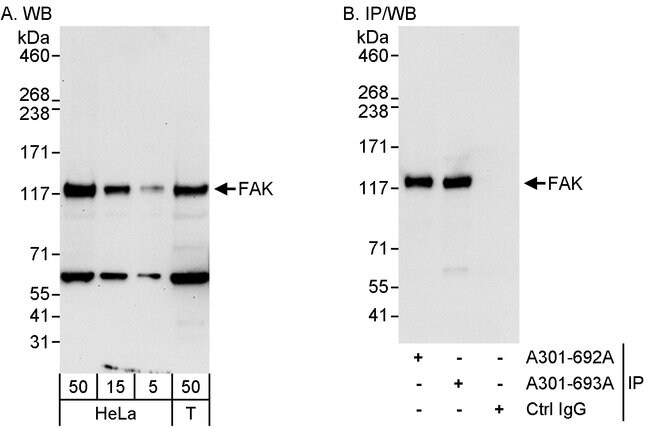
Image Source: Google
FAK is a catalytic central domain as well as a C-terminal tail that binds it to focal adhesions. These are places where cells connect to the extracellular matrix through the surface integrin receptors.
A rise in FAK Tyrosine phosphorylation happens after integrin interaction with fibronectin. The adhesion of murine NIH3T3 cells to fibronectin facilitates interaction with grb2.
Different transcript variants that encode various forms have been identified to be associated with FAK. FAK gene, however, the full-length characteristics of only three have been identified.
The Focal Adhesion Kinase (FAK) is one of the frequently expressed cytoplasmic proteins, a tyrosine kinase that is involved in the signal transduction process mediated by integrin.
It plays a crucial role in the regulation of various biological processes, which include cell spread, migration, and the survival of cells.
The activation of FAK through integrin clustering results in autophosphorylation of Tyr397 which serves as an important binding site that is a binding site for Src family Kinases PI3K as well as PLCg (2-5).
Phospho-FAK (Tyr397) Antibody can detect endogenous concentrations of FAK only when phosphorylated to Tyr397. This antibody may cross-react with other tyrosine-phosphorylated RTKs.